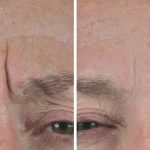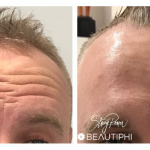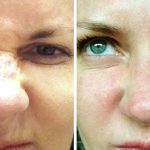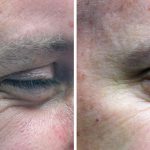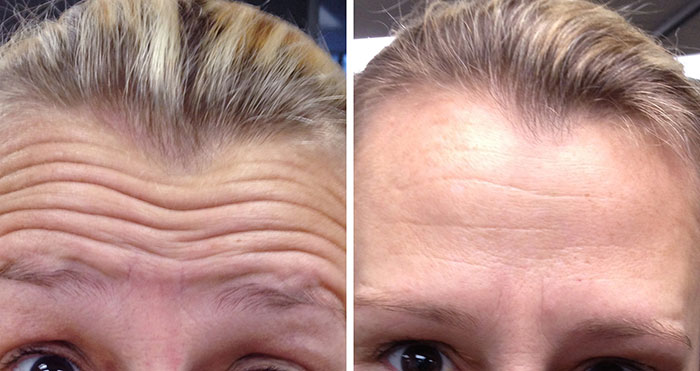
Treatment
Tiny injections to relax muscles
Recovery Time
10 minutes
Longevity
3-9 months
The 3 main botulinum toxins used in appearance medicine are introduced and outlined below.
What is Botox?
Botox ® is a natural, purified protein that relaxes wrinkle-causing muscles, creating a rejuvenated appearance. The active ingredient in Botox ® treatment is derived from bacteria in much the same way penicillin is derived from mould. It is manufactured in sterile laboratory conditions.
Botox ® is a prescription-only medicine and treatment is only available from a qualified medical professional. It is registered for the treatment of crow’s feet (the lines that radiate from the corner of your eye when you smile or squint), forehead lines (the horizontal lines that form when you raise your eyebrows) and glabellar frown lines (the vertical lines that appear between your eyebrows when you frown).
How BOTOX Cosmetic Works
Under normal conditions, a neurotransmitter known as acetylcholine is released by the nerve cell. Acetylcholine vesicles must be able to bind to the nerve membrane, in order to release the acetylcholine and transmit a signal to the muscle. Once released, acetylcholine triggers the muscle to contract. As you know, this repeated muscle contraction causes facial lines and wrinkles to form.
Once BOTOX Cosmetic is injected into the treatment area; the toxin enters the nerve and slices a special protein along the nerve membrane that is necessary for acetylcholine to bind properly. Because of this mechanism, acetylcholine cannot bind to the nerve membrane and is not released. Without the release of the neurotransmitter, acetylcholine, facial muscles are unable to contract and are temporarily paralyzed, giving the face a smooth, less wrinkled appearance.
BOTOX Cosmetic Candidates
Ideal candidates for BOTOX treatments are those that wish to see an improvement in their facial wrinkles and lines, however; they do not want to commit to facial plastic surgery just yet. Pregnant women are advised to avoid treatment, as the effect on the fetus is not yet known, and patients with certain neurological disorders are not considered good candidates. BOTOX has been tested successfully in patients (male and female) between the ages of 18 to 65 years old.
Procedure Preparation
Prior to your procedure, the treatment area will be thoroughly cleansed, usually with an alcohol-based cleanser.
BOTOX Cosmetic is injected into the specific muscle that is contracting and causing the lines and wrinkles on the face with a very fine needle. Treatment is effective, safe, and takes approximately 10 minutes.
The pain is a minimal “pinprick” and no anesthetic is required normally; however, some physicians will give you an option of using a topical anesthetic treatment, such as EMLA Cream to help you stay comfortable and relaxed during the procedure. If an anesthetic has not been administered, you may feel a slight stinging or burning sensation during the procedure.
Before performing the injections, the physician will ask you to contract your facial muscles in the desired treatment area in order to determine the precise injection sites. The entire procedure typically takes approximately ten to fifteen minutes but may vary depending on the number of desired treatment areas.
Factors such as age, sun exposure and or course heredity all contribute to the primary causes of facial lines and wrinkles. Also repetitive muscle contractions, such as squinting from the sun; can also contribute to the formation of wrinkles.
Nerves release neurotransmitters, which are chemicals that send signals, or impulses to other nerves, muscles, or the brain. Nerve impulses are what trigger muscles to contract or move. When injected into facial muscles, the toxin in BOTOX Cosmetic injections blocks the transmission of these nerve impulses, which temporarily paralyzes muscles and prevents them from contracting.
What to expect during your BOTOX Treatment
BOTOX Procedure FAST FACTS:
The procedure takes 5 minutes, and you can return to work or daily activities immediately.
Your doctor/nurse may ask you to squeeze and relax the muscles that were injected several times during the first day to help disperse the BOTOX.
Some people experience a slight twitching of the muscles as the BOTOX begins to do its work relaxing those muscles.
Some patients experience a mild headache for the first few days also.
Some may notice drooping of their upper eyelid or eyelids. This is due to BOTOX affecting the muscle that opens the eyelid. If this happens, your doctor/nurse can ensure an eye-drop can be prescribed that will help open your eyelid until that effect wears off. With an experienced injector, you should not have this happen.
Results of BOTOX Cosmetic injections are noticeable in a few days and last for several months.
Botox® is used to treat:
- Frown lines
- Crow’s feet
- Bunny lines
- Grumpy face lines (DAO’s)
- Excessive Sweating
Recovery from BOTOX Cosmetic Treatment
Recovery following BOTOX treatment is very minimal and brief. You will be able to resume your normal activities right after the injections. The only issue that is recommended is that you do not lie down immediately following your procedure. No massaging should take place in the area that was treated, along with no strenuous activities for the first 24 hours.
Botox FAQs
When can you start seeing the effects of Botox®?
After the injection, it takes 2-3 days for the treatment to begin taking effect, and about 7 days to see the full effect. The dose recommended for the treatment will vary depending on your individual situation and the severity of your lines. Your medical professional will know what the best dose is for you, and which muscles to inject to give you the best results.
How long do the effects of Botox® last?
The effects can last up to 4 months before lines slowly begin to return. If you have repeated treatments, the effect tends to last longer.
Difference Between BOTOX and Facial Fillers
BOTOX Cosmetic temporarily corrects or eliminates the dynamic wrinkles due to overactive facial muscles by blocking the chemicals that cause the facial muscles to contract to result in muscle relaxation. BOTOX is only effective for the correction of dynamic wrinkles. Skin sagging and wrinkles of the face start to develop as we get older; they are the result of thinned and broken collagen.
These types of wrinkles are different from the dynamic wrinkles and require the use of fillers to fill out and plump up the depressed areas. The intradermal injection of the filler masks the wrinkle by adding volume.
There are several available fillers; they include:
- Juvederm
- Restylane
- Anteis
- Radiesse
What are Advantages Of BOTOX
- The whole procedure takes little time, no anesthesia is needed and the person can get back to his/her usual activities almost immediately
- The effects appear in a short period of time (3 to 5 days) and last from three to four months
- The treatment can be repeated and in time its effects will start lasting longer
- It can also help with sweating (by paralyzing the muscles responsible for it) when injected in small amounts into the underarm skin or skin on the palms and soles of the hand or foot
- Improves the appearance of the skin
- BOTOX relieves migraine headaches
What are Disadvantages Of BOTOX
- Pain and bruising or bleeding at the injection site
- Temporary headaches can occur
- Results are temporary – last 3 to 6 months
- Treatment can be expensive over the long term – costs vary
- Common side effects include mild to moderate headaches, flu symptoms, temporary eyelid drooping and nausea
- Less than 1% of patients have reported the drooping of the eyelid.
- Unnatural facial expressions due to the certain parts of the face being blocked with BOTOX
- Some believe that because some parts of the face are “frozen”; that the brain redirects the impulses to other muscles and wrinkles can grow in parts of the face which are not being treated
- BOTOX should never be used on women who are pregnant
Does Botox® cause more wrinkles to form?
Botox ® will relax the facial muscles that cause expression lines. It does not cause more wrinkles to form. If you choose not to have further treatments, your muscles will eventually return to normal activity, and your wrinkles will gradually revert to their pre-treatment levels.
Will facial expression be affected by Botox®?
Botox ® works specifically on the muscles treated. This means that any muscle left untreated, including untreated facial muscles, should not be affected. Your medical professional will determine the best dose for you, and which muscles to inject to give you the best results.
Are there any side effects of Botox®?
In addition to its use for cosmetic reasons, Botox ® is also used to treat patients in a variety of therapeutic indications, including cerebral palsy in children, movement disorders (head, neck, shoulder, face), and axillary hyperhidrosis (excessive underarm sweating).
In the amounts used for the treatment of upper facial lines, the side effects are usually temporary and localized to the area of injection. Possible side effects include headaches, pain, burning/stinging, bruising, swelling or redness at the injection site, local muscle weakness, including drooping eyelids/eyebrows, swollen eyelids, skin tightness, tingling sensations, itching/aching forehead, nausea, and flu-like symptoms.
How long has Botox® been used?
Botox ® has been used in Australia and New Zealand in therapeutic treatments for over 30 years. It was first approved for use in New Zealand in 1991.
Can Botox® be combined with other products and procedures?
Many people use a combination of cosmetic products and/or procedures with Botox ®. Your medical professional will advise you whether you will receive better results if your Botox ® treatment is combined with other products and/or procedures.
Other forms of Botulinum Toxin – Dysport® and Xeomin®
What is Dysport®?
Dysport® is a simple, effective, non-surgical treatment that works by relaxing facial muscles, thereby reducing and smoothing away frown-lines and wrinkles. Dysport®, like Botox®, is a protein extracted from the bacterium Clostridium Botulinum. The Dysport® protein was initially used for the treatment of motor disorders and various kinds of involuntary muscular spasms, including cerebral palsy.
How does Dysport® work?
Dysport® blocks impulses from the nerves to the facial muscles that are related to expression lines. Dysport® relaxes the muscles so they do not contract. After treatment, the overlying skin becomes smooth and unwrinkled, while the untreated facial muscles contract in a normal fashion, allowing normal facial expression to be unaffected.
Dysport® injections have been used safely and effectively for over fifteen years to treat many ophthalmologic and neurological disorders. Numerous safety studies performed around the world have shown it to have an equal or more favourable safety profile than Botox®.
History of Dysport®
Though approved in the United States in April 2009, Dysport® has a history of use in other countries since 1991. Dysport® was first approved for aesthetic use outside the US in 2001, and has steadily gained in popularity around the world.
Dysport® was developed in the United Kingdom in the early 1990s to successfully treat a number of neurological and ophthalmic conditions. It was further developed to treat a wide variety of neuromuscular disorders, for which it is licensed in over 60 countries.
Since that time, with an increased understanding of the uses of Dysport®, thousands of treatments have been safely and effectively performed for a variety of conditions ranging from facial wrinkles to axillary hyperhidrosis (excessive sweating under the armpits).
Today, Dysport® is marketed and sold for aesthetic use in many countries around the world and is approved by the US Food and Drug Administration.
What is Xeomin®?
Xeomin® is the purest form of Botulinum Toxin on the market today. It does not have the complexing proteins that its predecessors have, which can be one of the factors involved in treatment failure with the other forms of Botulinum Toxin.
In other words, if you are treated with this toxin only, you will have less likelihood of a strong immune reaction. The less pure the toxin, the stronger the immune reaction that can be triggered. Botox® has more complexing proteins, Dysport® has less, and Xeomin® has none.
Like Dysport® and Botox®, Xeomin® works by paralyzing wrinkles. It blocks the signals from the nerves to the muscles. As a result, the targeted muscle cannot contract.
How is Xeomin® made?
In nature, C. botulinum produces the toxin in association with accessory proteins (toxin complex). Merz takes the toxin complex and employs a proprietary manufacturing process that isolates the therapeutic component and removes the accessory proteins to produce Xeomin®.
More FAQs
Are all Botulinum Toxin products the same?
Not all Botulinum Toxin products are the same. This is reflected in the unique names for each product. The dose of Xeomin® may be different from other Botulinum Toxin products. If you are interested in Xeomin® injections, or wish to discuss the different Botulimum Toxins, and which one is right for you, feel free to make a consultation with one of our Cosmetic Medicine Nurses.
Xeomin® versus Botox® and Dysport®
Botox®, Dysport®, and Xeomin® have a lot in common, but they also have some important differences. Unlike its predecessors, Xeomin® does not need to be refrigerated. This may be an advantage when it comes to distribution. What’s more, Xeomin® is “naked”. There are no additives – just Botulinum Toxin Type A. This may lessen a patient’s likelihood for developing antibodies to Xeomin®. When your body senses a foreign invader, it responds by creating antibodies and launching an attack. If this were to occur with a neurotoxin such as Botulinum Toxin Type A, then it may not be as effective as planned.
Xeomin® is said to be more like Botox® than Dysport®. It takes about one week for the full effects of Xeomin ®injections to be realised, and once this occurs, the results last from three to six months.